Urea - Guide to Trigger the Growth of Fertilizers Industry in the Coming Years
Urea is produced with a combination of carbon dioxide and ammonia. It is a highly-efficient nitrogen fertilizer used in agriculture for ground treatment and top-dressing and is also used for all soil types and crop plants. Urea significantly increases agricultural yield. In the industry application, it is used for the production of plastics, glues and resins. Urea also finds application as ruminant's forage additive agent. Urea is used as a raw material for the manufacture materials such as urea-formaldehyde resins and urea-melamine-formaldehyde used in marine plywood.
Urea is the world's most commonly used nitrogen fertilizer and indeed more urea is manufactured by mass than any other organic chemical. Containing 46% N, it is the most concentrated nitrogen fertilizer, and is readily available as free-flowing prills (granules). It is the cheapest form of nitrogen fertilizer to transport and it is also the least likely to 'cake'. It is therefore favoured in developing countries.
While over 90% of urea produced is used as a fertilizer, it has other uses, which include the manufacture of the melamine, used in melamine-methanal resins. Urea itself also forms important resins.
An increasingly important use of urea is in reducing air pollution from diesel engines in cars, buses and lorries. Diesel engines run at high temperatures and nitrogen and oxygen, from the air, are able to react together under these conditions to produce high concentrations of nitric oxide. One way to remove this pollutant is to allow it to react with ammonia to form nitrogen.
However it is not possible to use ammonia directly as it is too volatile and is poisonous. Instead a solution of urea in water is injected into the hot gases emerging from the engine in the exhaust. Urea is thermally decomposed to ammonia and carbon dioxide.
Manufacture of Urea:
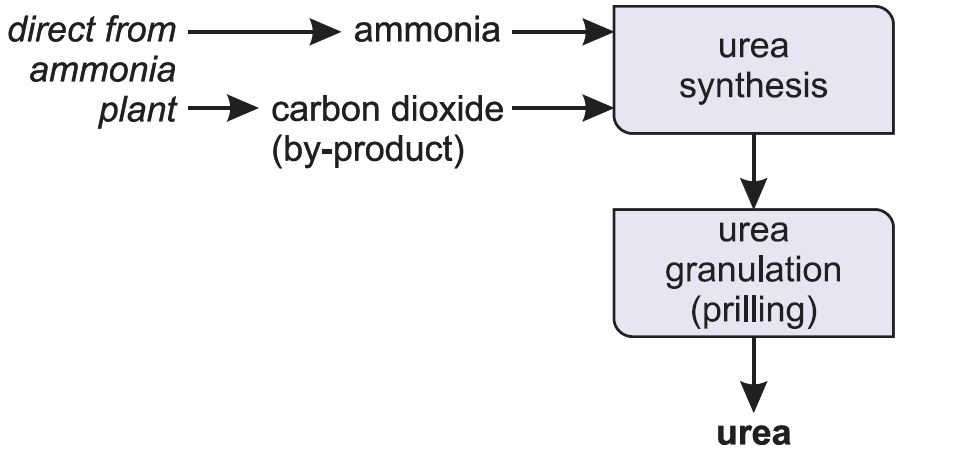
Ammonia reacts with carbon dioxide to produce urea. Urea is always manufactured close to an ammonia plant (Figure 5).
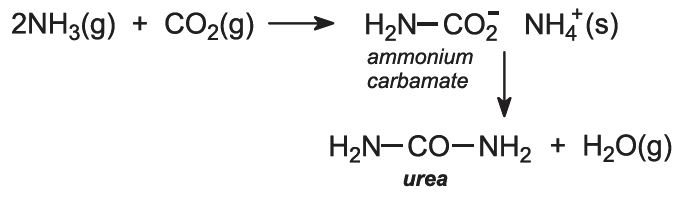
Ammonia and carbon dioxide are heated together at 450 K and 200 atm pressure. First ammonium carbamate is formed, which rapidly decomposes to form urea:
Urea is the world's most commonly used nitrogen fertilizer and indeed more urea is manufactured by mass than any other organic chemical. Containing 46% N, it is the most concentrated nitrogen fertilizer, and is readily available as free-flowing prills (granules). It is the cheapest form of nitrogen fertilizer to transport and it is also the least likely to 'cake'. It is therefore favoured in developing countries.
While over 90% of urea produced is used as a fertilizer, it has other uses, which include the manufacture of the melamine, used in melamine-methanal resins. Urea itself also forms important resins.
An increasingly important use of urea is in reducing air pollution from diesel engines in cars, buses and lorries. Diesel engines run at high temperatures and nitrogen and oxygen, from the air, are able to react together under these conditions to produce high concentrations of nitric oxide. One way to remove this pollutant is to allow it to react with ammonia to form nitrogen.
However it is not possible to use ammonia directly as it is too volatile and is poisonous. Instead a solution of urea in water is injected into the hot gases emerging from the engine in the exhaust. Urea is thermally decomposed to ammonia and carbon dioxide.
Manufacture of Urea:
Ammonia reacts with carbon dioxide to produce urea. Urea is always manufactured close to an ammonia plant (Figure 5).

Ammonia and carbon dioxide are heated together at 450 K and 200 atm pressure. First ammonium carbamate is formed, which rapidly decomposes to form urea:





Comments
Post a Comment